Platinum
Product Details
-
Platinum has excellent performance and it is widely used in many areas. In the jewelry industry, it is mainly used for ornaments and handicrafts. In the chemical industry, it is made into catalysts for advanced chemical vessels, platinum crucibles, electrodes and accelerated chemical reactions. Platinum-iridium alloy is also the material for fountain pen nibs.
-
The color and luster of platinum is natural.The hardness is 4 ~ 4.5 degrees,the relative density is 21.45, and the proportion is 15 ~ 19 or 21.4.The ductility of Platinum is strong, and the metal can be drawn into very thin platinum wire and rolled into extremely thin platinum foil. Its strength and toughness are much higher than those of other precious metals. A gram of platinum can be extended to 1.6 kilometers in length without breaking. The melting point of platinum is 1773.5 ℃. Its thermal conductivity is good. The chemical properties are extremely stable, insoluble in strong acid and strong alkali, and do not oxidize in the air. Platinum does not absorb mercury and has a unique catalytic effect.
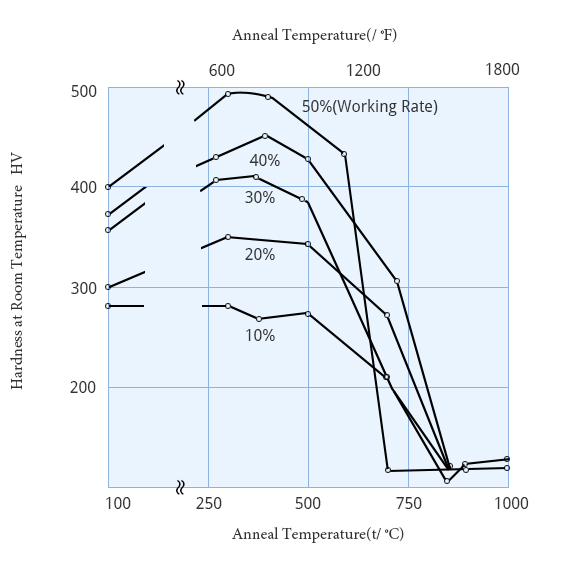 Hardness of 99.9%purity platinum after rolling and annealing(anneal time 1h)
Hardness of 99.9%purity platinum after rolling and annealing(anneal time 1h)
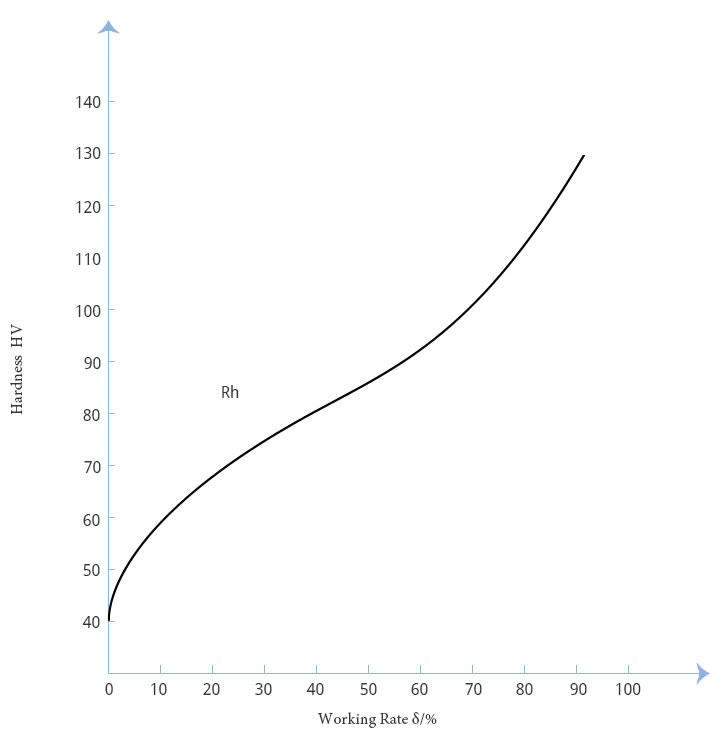 The hardening process curve of high purity platinum
The hardening process curve of high purity platinum
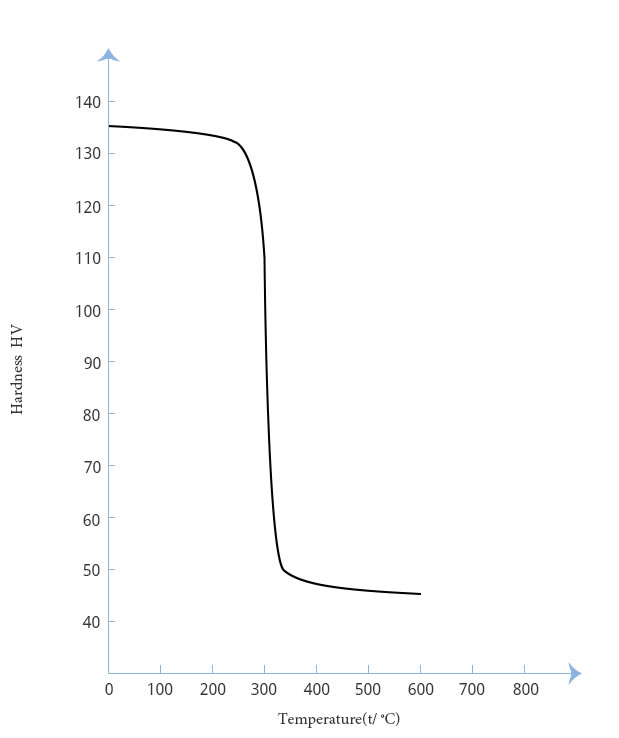 The annealing curve of high purity platinum
The annealing curve of high purity platinum






